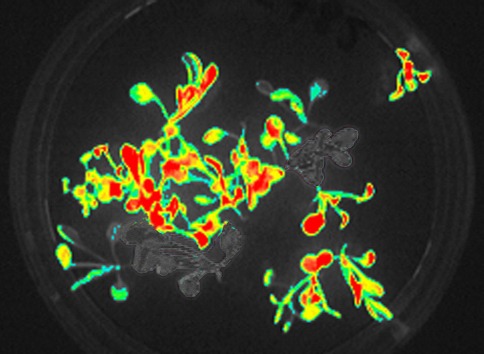
The Z125 GFP Closed FluorCam is designed for both chlorophyll fluorescence and other fluorescent protein imaging. The basic system of the Z100 FluorCam – Closed (CCD camera, four fixed LED panels) is supplemented here by a fully motorized and software-controlled filter wheel and appropriate filter sets for the detection and imaging of green fluorescent protein (GFP) in the standard system, red-shifted GFP, or yellow fluorescent protein (YFP) (optional). The blue emitting and red fluorescent protein can be detected and imaged in absence of chlorophyll. The device configuration also provides a user-friendly and quick transition between the GFP and chlorophyll fluorescence detection and imaging.
The Z125 GFP FluorCam – Closed System similarly to the Z100 FluorCam – Closed System consists of a high resolution and long integration time TOMI-2 CCD camera (1360×1024 pixels), 4 fixed LED panels that illuminate the sample, imaging software with protocols and 7 position filter wheel for imaging chlorophyll and GFP. Filters for other fluorescent proteins are optional. The LED panels provide uniform irradiance over samples up to 10 x 10 cm – suitable for small plants, detached leaves, petri plates with seedlings or algal cultures, etc. The system allows dark adaptation and includes a high-performance PC and a comprehensive software package for control of the system, data acquisition and analysis.
- Transient GFP Fluorescence
- Fv/Fm
- Quenching.
- Kautsky induction.
- QA reoxidation (needs optional accessories).
- OJIP (needs optional accessories).
- Fast fluorescence induction with 1µs resolution (needs optional accessories).
for details of chlorophyll fluorescence parameters measured by this system see Z100 FluorCam – Closed
- Imaging of chlorophyll fluorescence and GFP fluorescence on the same tissues.
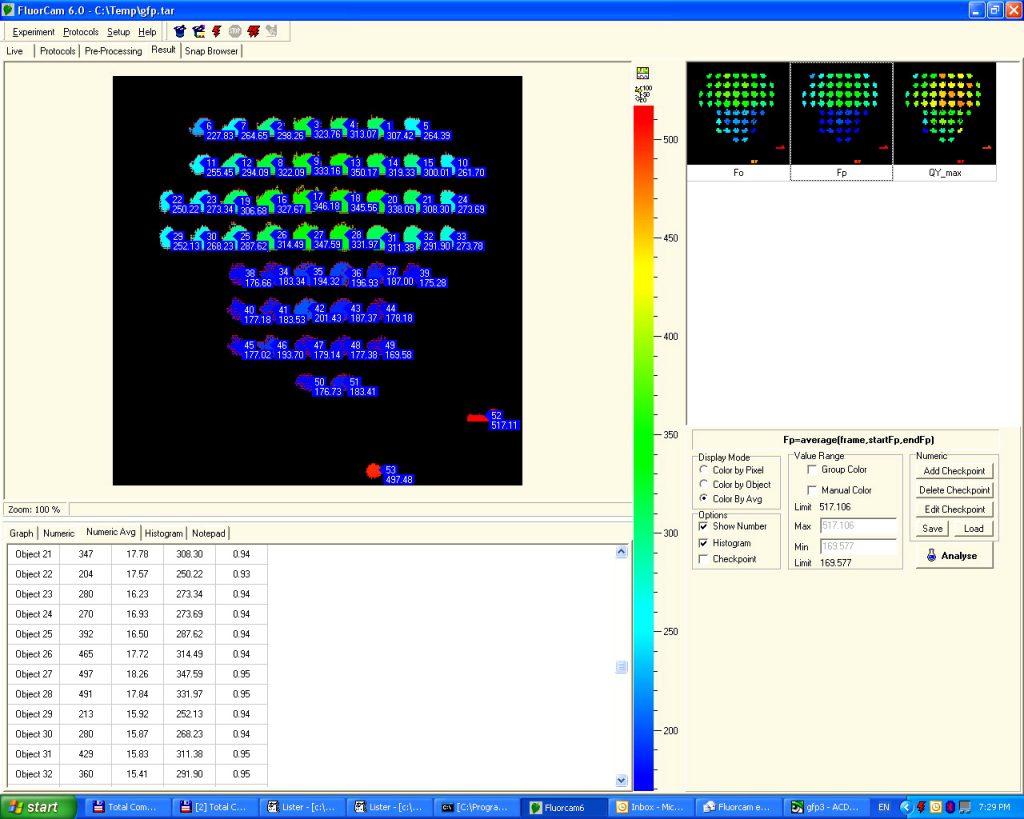
- Included high-resolution, long integration CCD camera: full resolution 1360 x 1024 (20 fps), operates in video or snapshot mode
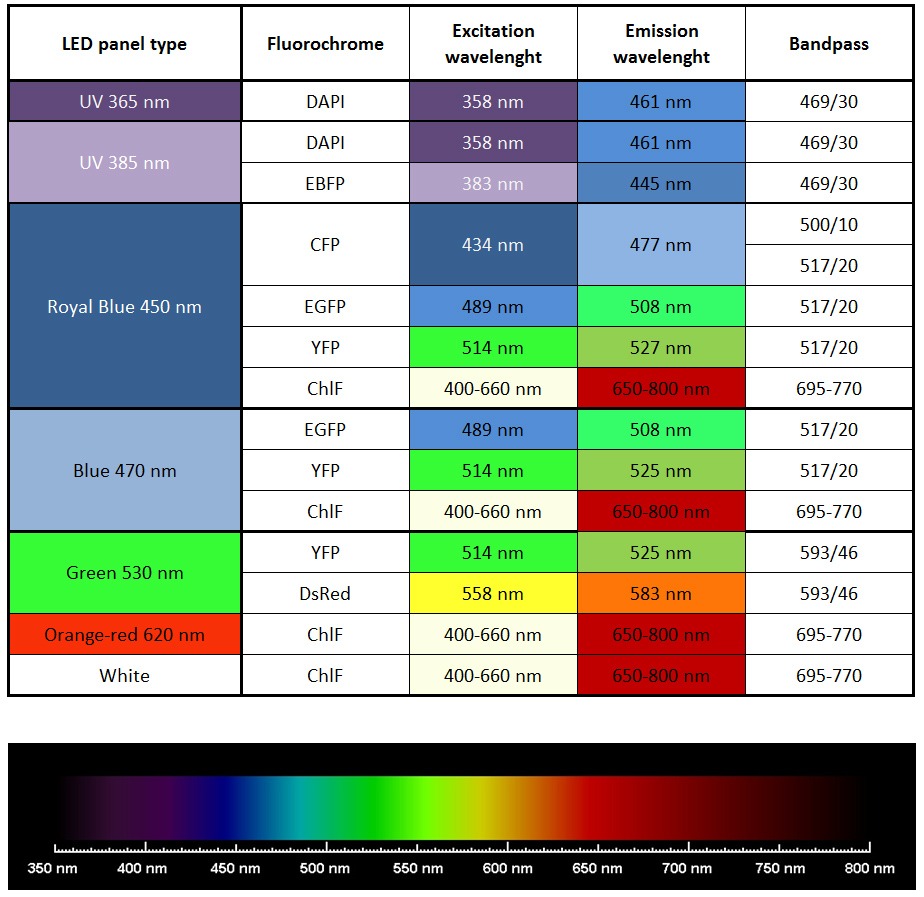
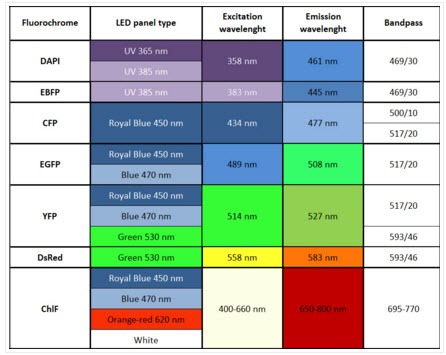
- Four light-upgraded LED panels (4 panels 470 nm OR 2 panels 447 nm + 2 panels 470 nm OR 2 panels 627 nm and 2 panels 470 nm) Additional UV, 505, 570, 605, 630, 735 nm (optional).
- 7 position filter wheel – motorized and software-controlled (included).
- Adjustable shelf system for different plant sizes (included).
- Dark chamber for adaptation (included).
- Supplied with a high-end computer and user-friendly FluorCam software (included).
- Chlorophyll Fluorescence Imaging: Plants and Algae
- GFP Detection and Expression Studies (Plants and Animal Tissues)
- Biotic and Abiotic Stress Resistance in Plants
- Plant Pathogen Research
- Plant Microbe Interaction
- Bacterial – Protozoan Interaction
- Chl Fluorescence parameters: (F0, FM, FV, F0’, FM’, FV’, QY(II)), Abs PAR-value, or the parameters that are calculated from fluorescence emission (e.g., NPQ, FV/FM, FV’/FM’, Rfd, qN, qP), PAR-absorptivity, photosynthetic electron transport rate (PS), and others
- Excitation light sources: Standard: red-orange (617 nm) and royal-blue (447 nm) or blue (470 nm),
- Saturating pulses intensity: 4,900 µmol(photons)/m²/s (in a standard version), 6,000 µmol(photons)/m²/s (in the light-upgraded version)
- Actinic light intensity: Up to 2,000 µmol(photons)/m²/s (in standard), up to 3,000 µmol(photons)/m²/s (in light upgraded version)
- Filter wheel: 7 positions (includes GFP and chl filters in standard system)
- Light regime: Static or dynamic (sinus form)
- Custom defined protocols: Variable timing, special language and scripts
- CCD detector wavelength range: 400 – 1000 nm
- CCD format: full resolution 13602 x 1024 (20 fps), two operation modes: video and snapshot
- Imaging frequency: Maximum 50 frames per second
- pixel size: 6.45 µm x 6.45 µm
- A/D resolution: 16 bit (65,536 grey levels)
- Spectral response: QE max at 540 nm (~70 %), 50 % roll-off at 400 nm and 650 nm
- Read-out noise: Less than 12 electrons RMS – typically only 10 electrons
- Full-well capacity: Greater than 70,000 electrons (unbinned)
- Bios: upgradeable firmware
- Communication port: USB 2.0
- Outer dimensions: 471 mm (W) x 473 mm (D) x 512 mm (H)
- Weight: 40 kg
- Power input: 1100 W
- Electrical: 90 -240V
-
BOURDAIS G., BURDIAK P., GAUTHIER A., ET AL. (2015) Large-Scale Phenomics Identifies Primary and Fine-Tuning Roles for CRKs in Responses Related to Oxidative Stress. PLoS Genet,. Volume 11.DOI:10.1371/journal.pgen.1005373
- Leal M.C. et al (2014): Marine Ecology (2014): 1-13, doi: 10.1111/maec.12164
-
BEIKE A. K., LANG D., ZIMMER A. D., ET AL. (2015). Insights from the cold transcriptome of Physcomitrella patens: global specialization pattern of conserved transcriptional regulators and identification of orphan genes involved in cold acclimation. The New Phytologist. Volume 205, Pages 869-881.DOI:10.1111/nph.1004
-
VERCRUYSSEN L., TOGNETTI V.B., GONZALEZ N., ET AL. (2015). GROWTH REGULATING FACTOR5 Stimulates Arabidopsis Chloroplast Division, Photosynthesis, and Leaf Longevity. Plant Physiology. Volume 167, Pages 817 -32. DOI:10.1104/pp.114.256180