The Z150 Photobioreactor is used for precise phototrophic cultivation of algae and cyanobacteria. It features a unique combination of the cultivator and monitoring device. Light intensity and timing, spectral composition, and temperature can be set with high accuracy.
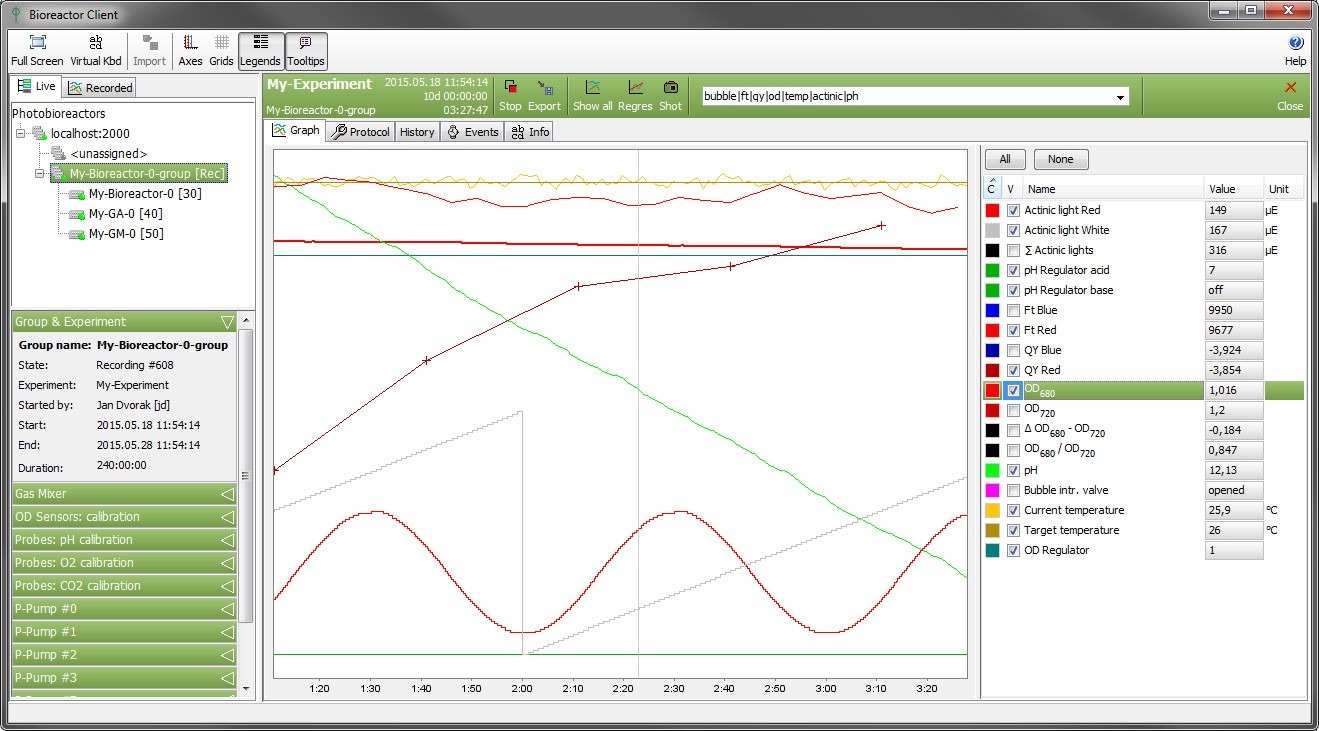
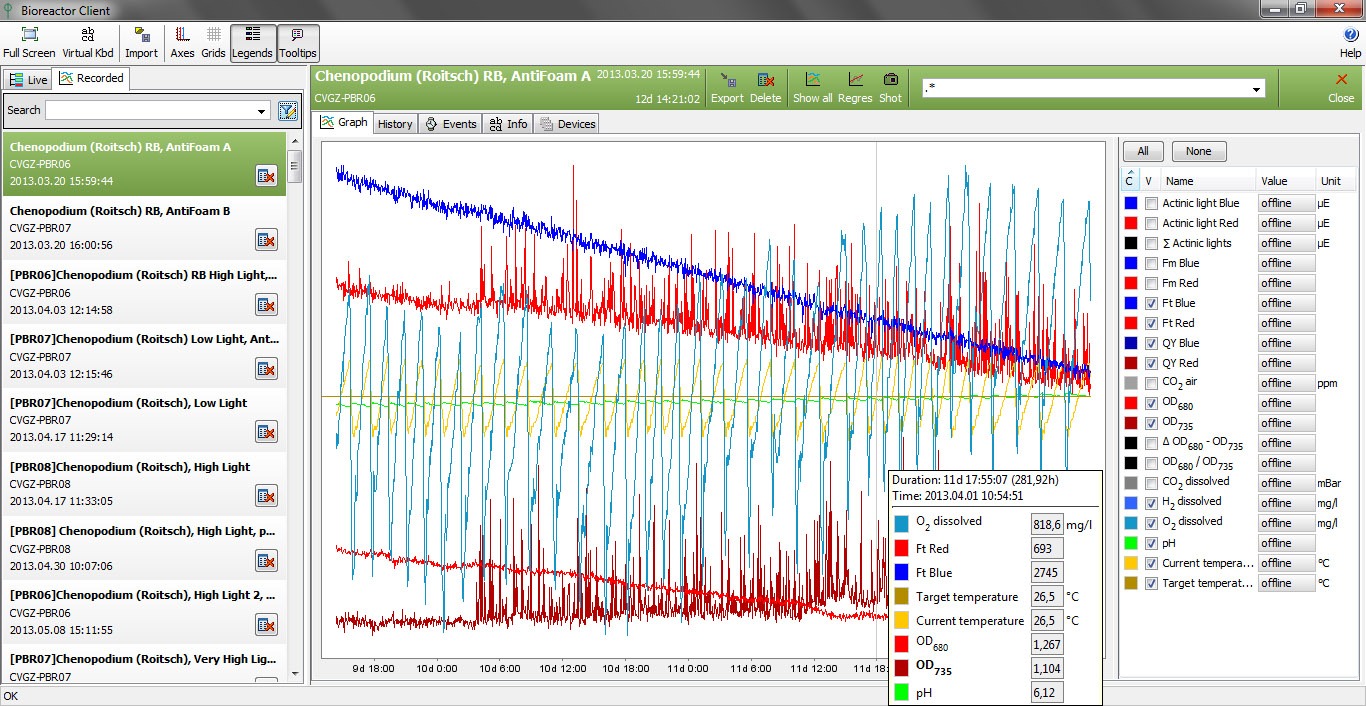
The cultivation conditions of a photobioreactor can be varied according to user-defined protocol. The growth of the algal culture is monitored by the integrated densitometer (OD720, OD680). The chlorophyll content of the culture can also be monitored continuously by the difference in optical densities at 680 and 720 nm. The instantaneous physiological state of the culture is measured by the Photosystem II quantum yield Fv/Fm (QY).
For cultivation of up to 8 different algal cultures please see the Z160 Multicultivator
- Cultivation vessel capacity: 400ml Standard, 1000ml (Z150-1L), 3000ml (Z150-3L), 25L (Z150-25L), 100L (Z150-100L)
- LED light illumination with full control of intensity (up to 1500uE standard or 3000uE enhanced), quality, duration, mode
- Temperature control 15-55C (standard) or 0-70C (enhanced)
- Continuous culture options (chemostat, turbidostat modules)
- Integrated Double-Modulation Fluorometer with online measurement of chlorophyll fluorescence parameters, such as Ft, QY (Fv/Fm) (standard)
- Integrated online optical density (720, 680nm) measurement (standard)
- Dissolve O2 and CO2 measurements (optional)
- pH measurements (standard) and control (optional)
- Control software with protocols and data collection.
- Remote online access to data and control
- Phototrophic cultivation and monitoring of algae and cyanobacteria
- Studies of algal and cyanobacteria photosynthesis
- Algal biomass and biofuel production in floor standing models (25, 100L)
- Fluorescence Parameters: Ft, Fv/Fm (QY) .
- Vessel Capacity: 400 ml, 1L, 3L, 25 L, 100 L
- Precision Controlled Temperature: 15 °C – 55 °C (standard), 0-70°C (optional)
- Cooling and Heating System: Two 200 W or 400W (Z150-3L) Peltier elements.
- Actinic and Saturating Light: Two standard versions: one with blue and red LEDs (450 nm, 630 nm); the other with white and red LEDs. Light intensity is adjustable from 0 ~ 1500 μmol(photon)/m2/s, with light upgrade 0 ~ 3000 μmol(photon)/m2/s. Other LEDcolors available as an option
- Two Displays: For system control and actual temperature.
- Light Regime: Static or dynamic (sinus, triangle etc).
- Optical Density Measurement: Real time measurement of OD by two IR LEDs (720 nm, 680 nm).
- Detector Wavelength Range: PIN photodiode with 697nm -750 nm bandpass filters.
- Controlled Flow and Composition of the Bubbling Gas: Optional.
- Pumps: Up to 8 peristaltic pumps supported (optional),
- pH/temp Probe included.
- Bios: Upgradeable firmware.
- Communication Port: USB
- Material: Glass, stainless steel, silicon gasket.
- Size: 50 x 25 x 35 cm (for the standard 400ml unit).
- Weight: 15.5 kg (400ml unit), 17.5kg (1L), 28kg (3L).
- Electrical: 90-240 V.
- ČERVENÝ J., SINETOVA M. A., ZAVŘEL T. AND LOS D.A. (2015): Mechanisms of
High Temperature Resistance of Synechocystis sp. PCC 6803:
An Impact of Histidine Kinase 34. Life. Volume 5,
Pages 676-699. DOI:10.3390/life5010676 - PROKOP A., BAJPAI R. K. AND ZAPPI M. E. (2015): Algal Biorefineries: Volume 2: Products and Refinery Design. Springer, Pages 557. DOI:10.1007/978-3-319-20200-6
- VAN ALPHEN P. AND HELLINGWERF K. J. (2015): Sustained Circadian Rhythms in Continuous Light in Synechocystis sp. PCC6803 Growing in a Well-Controlled Photobioreactor. PLoS ONE 10. DOI:10.1371/journal.pone.0127715
- PAGE L. E., LIBERTON M., AND PAKRASI H. B. (2014): Reduction of Photoautotrophic Productivity in the Cyanobacterium Synechocystis sp. Strain PCC 6803 by Phycobilisome Antenna Truncation. Applied and Environmental Microbiology. Volume 78, Pages 6349–6351. DOI:10.1128/AEM.00499-12
- ČERVENÝ J., SINETOVA M. A., VALLEDOR L., ET AL. (2013): Ultradian metabolic rhythm in the diazotrophic cyanobacterium Cyanothece sp. ATCC 51142. PNAS, Volume 110, Pages 13210–13215. DOI: 10.1073/pnas.1301171110
- VEJRAZKA C., JANSSEN M., BENVENUTI G., ET AL. (2013): Photosynthetic efficiency and oxygen evolution of Chlamydomonas reinhardtii under continuous and flashing light. Applied Microbiology and Biotechnology. Volume 97, Pages 1523–1532. DOI 10.1007/s00253-012-4390-8
- CARRIERI D., PADDOCK T., MANESS P. CH., ET AL. (2012): Photo-catalytic conversion of carbon dioxide to organic acids by a recombinant cyanobacterium in capable of glykogen storage. Electronic Supplementary Material (ESI) for Energy & Environmental Science
Software
Basic Version of the Software:
Online software upgrades, Calibrations of accessories, make changes to protocol duringexperiments, restore running experiments after laptop reboot, raw sensor data recording, logging of custom/system experiment events, export of data to MS Excel/LibreOffice, filtering of recorded experiments, management of users, support of Turbidostat and Chemostatprotocols, support of external lights protocol PWM pump or Stirrer protocol, measurements of individual Ft/QY parameters
Advanced Version of the Software:
All features of the BASIC edition are included plus Email notifications from the software (low level device commands), Scripting support in protocol (customization of protocols), customization of measuring periods in protocol, export and import of protocol cycles through clipboard, import of experiments in previous software version, scheduling of experiments, advanced regressions in experiment graph, support of additional peristaltic pumps